2023.01.23
[Paper] “Crystal Structures of Ce(IV) Nitrates with Bis(2-pyrrolidone) Linker Molecules Deposited from Aqueous Solutions with Different HNO3 Concentrations” published by WRH professors Satoru Tsushima and Koichiro Takao
WRH Specially Appointed Associate Professor Satoru Tsushima, Juliane März (Helmholtz-Zentrum Dresden-Rossendorf), Associate Professor Koichiro Takao (Institute of Innovative Research), et al. published a paper in Inorganic Chemistry.
The work is also the result of a Grant-in-Aid for Scientific Research B that the researchers were awarded.
https://doi.org/10.1021/acs.inorgchem.2c03554
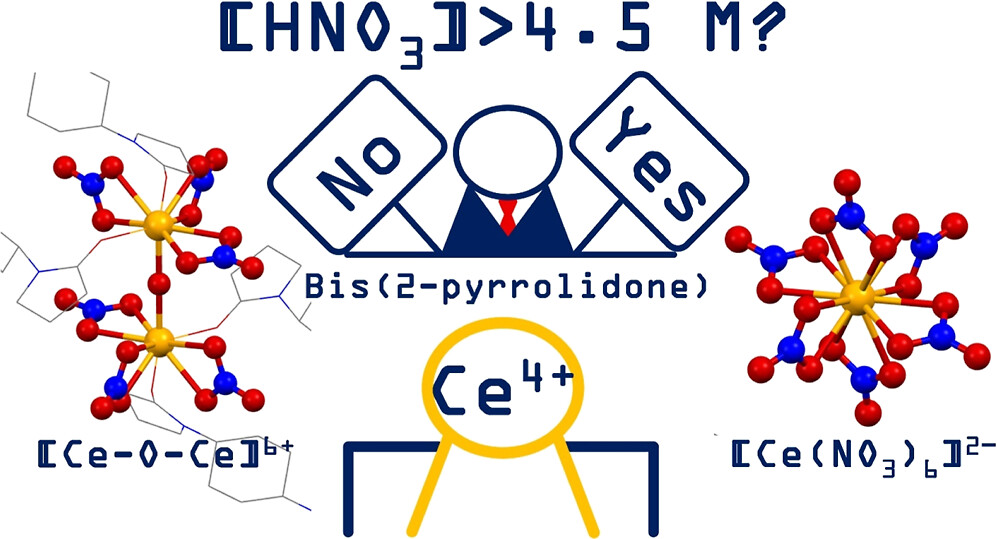
Abstract
We investigated the molecular and crystal structures of Ce(IV) compounds deposited under different [HNO3] with bis(2-pyrrolidone) linker molecules having a trans-1,4-cyclohexyl bridging moiety (L). As a result, we found that, after loading L, Ce(IV) in HNO3(aq) exclusively provides one of different crystalline phases, (HL)2[Ce(NO3)6] or [Ce2(μ-O)-(NO3)6(L)2]n 2D MOF, depending on [HNO3]. The former has been obtained at [HNO3] = 4.70–9.00 M and is isomorphous with the analogous (HL)2[An(NO3)6] we reported previously. In contrast, the deposition of the latter phase at the lower [HNO3] conditions (1.00–4.30 M) demonstrates that hydrolysis and oxolation of Ce4+ proceed even below pH 0 to provide a [Ce–O–Ce]6+ unit included in this compound. These different Ce(IV) phases are exchangeable with each other under soaking in HNO3(aq), implying that chemical equilibria of dissolution/deposition of these crystalline phases and hydrolysis and oxolation of Ce4+ and its complexation with NO3– occur in parallel. Indeed, such coordination chemistry of Ce(IV) in HNO3(aq) was well corroborated by 17O NMR, Raman, and IR spectroscopy.

